
A Review of Research on the Biological Transmutation of Chemical Elements
Based on an unfinished paper by Prof. L.W.J. Holleman
7. Holleman's Experiments 1975 - 1982
7.1. Materials and Methods
7.1.1. Overview
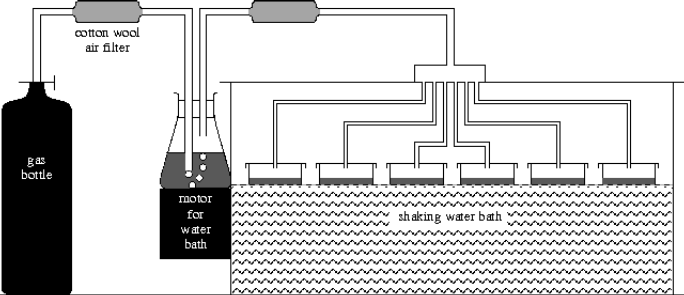
Figure 1.
The experimental set up is shown in figure 1. The culture methods used were those of standard laboratory practice. Thus bottled carbon dioxide gas was used to provide an air mixture containing 5% carbon dioxide. This was bubbled through a wash bottle that doubled up to grow the inoculation culture. This inoculation culture was used for the following cycle of the experiment. The gas was filtered by passing it through wads of cotton wool on both sides of the wash bottle and then split equally to pass over all six experimental cultures. The cultures were inoculated by adding 2ml of the previously grown inoculation culture to 50ml of a slightly modified standard chemical nutrient solution developed for the culture of the alga Chlorella vulgaris. The six experimental culture dishes were agitated and kept at a constant 27 degrees in a thermostatically controlled shaking water bath. The water bath contained twice distilled water and was provided with a loosely fitting lid, as were the culture dishes. Extra illumination was provided by a standard household 150W tungsten lamp suspended above the shaking bath.
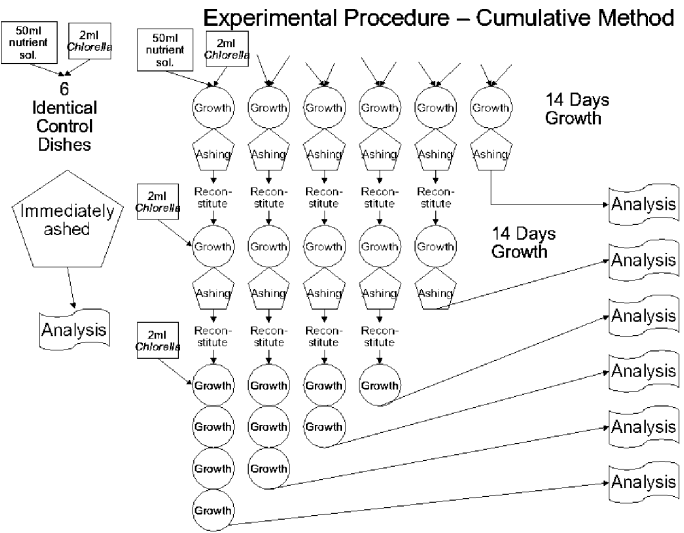
Figure 2.
The procedure is sketched out in figure 2 for the six experimental culture dishes. As stated above, all six experimental culture dishes began with 50ml of new nutrient solution and were inoculated with 2ml of a previously prepared culture solution. They were allowed to grow until the algae appeared to have visibly reached the full extent of their normal growth (or shortly after). This was about 12 days. The dishes, with their algae, were removed, evaporated and fully ashed in a furnace at 500 degrees. The ashes, still in their original dishes, were redissolved in distilled water to form the nutrient solution for the next cycle of the experiment. This was inoculated with 2ml of the culture grown in the gas wash bottle during the previous cycle.
The amounts of the chemical elements potassium and sodium were measured during the course of the experiment by the removal, at each cycle, of one of the experimental dishes after the ashing of its contents. Thus the first cycle started with six growth cultures, the second with five, through to the sixth and last cycle which consisted only of the one remaining dish.
The control cultures differed from the experimental ones in the first significant experiment of Holleman's (experiment II) on two points: 1. After adding the 2ml inoculation culture they were immediately ashed: 2. The dishes containing the ashes were kept in a desiccating chamber separate from the experimental cultures during the twelve days or so that the latter were growing. Only once the experimental dishes were themselves ashed were they and the controls that went with them kept together.
The controls of Holleman's third experiment (III) were modified to reduce the difference between the control and experimental dishes to just one point: the timing of an addition of 10ml of 0.01N nitric acid (HNO3). Rather than adding the acid during the ashing process, it was added for the controls as a component of the nutrient solution with the intention of making the nutrient solution unsuitable for the growth of Chlorella. This proved, however, to be unsuccessful.
7.1.2. Apparatus and Materials
7.1.2.1. Shaking Bath
For the homogeneous development of the algae an agitation of the surrounding fluid is necessary. This prevents clumping and maintains a suspension of single cells, thus ensuring an even supply of nutrient solution to all cells. This was attained by the culture dishes being shaken. The apparatus used for this purpose (Dubnoff Incu-Shaker, Labline model 61) was thermostatically controlled and was held at 27 degrees throughout the growth stages of the experiments. It shook to and fro at a frequency of 1 beat per second and an amplitude of 3.5cm. The dishes stood on a perforated plate and were held in place by screws fixed (with difficulty [!]) into the plate. The bath was filled to the bottom of the dishes with distilled water. The whole machine was completely covered by a Perspex lid made with a hole for the bringing in of carbon dioxide rich air. Although during the experiment water condensed out onto the lid, it appeared that evaporation from the experimental dishes was mostly acceptable as their volume by the end of a growth period was practically unchanged.
7.1.2.2. Containment Materials
The algal suspension was contained in round quartz glass dishes with straight vertical sides (diameter 10cm, height 1.5cm, thickness 0.3cm). The material, Vitreosil, withstands treatment with acids and heating to 500 degrees extremely well. Before being used the dishes were thoroughly cleaned with an alkali scouring material (Vim) and rinsed for a prolonged period of time in deionised water, twice distilled.
7.1.2.3. Other Glassware
The provision of solutions and the culture of algae occurred in standard, cleaned laboratory glassware (Jena G... [?]).
The measuring glassware used (pipettes, measuring cylinders) were calibrated. Moreover, for each specific handling, one and the same item (pipette, measuring cylinder) was used with special consideration for the identical handling of proof and control.
The stock nutrient solution and the ash solutions that were obtained were stored in good closing polythene bottles.
7.1.2.4. Chemicals
With all work, Merk analytical chemicals were used. In particular, nitric acid, which was used in fairly large quantities in the ashing and reconstituting of the nutrient medium, was in this regard, critical. A proportion was isolated for direct use from a measured quantity (1 litre) of concentrated HNO3 intended for this work.
7.1.2.5. Gas Supply
Algal growth was promoted by the provision of a carbon dioxide rich atmosphere. The CO2 was lead from a gas cylinder, through a control valve and then passed through a wash flask (held at 27 degrees) which contained nutrient solution and which was inoculated with an algal suspension with the same content as the experimental cultures. This solution acted as inoculation material for the following growth period. The gas stream on either side of the wash flask was filtered by a wad of cotton wool.
The CO2 enriched air passed on through a connecting tube of silicon rubber into a dividing head. This was situated in the Perspex lid of the shaking bath and served to distribute the gas stream equally, via 6 identical lengths of rubber tubing to each of the six culture dishes, exiting through their Perspex lids into the air space above the culture liquid. The underside of these lids was furnished with three ribs whereby a space of approximately 1mm above the walls of the dishes allowed the gas mixture to escape.
7.1.2.6 Lighting
The necessary provision of radiant energy [light!!] was met by use of a 150 Watt Philips lamp [almost certainly a household tungsten bulb], 50cm above the experimental set up. A rough estimate of the light intensity was 1000-1500 Lux.
Lighting, gas flow and shaking bath were generally only switched on during working hours [10am-5pm Monday to Friday]. On only a few occasions did this handling continue through the night.
7.1.2.7 Sterilisation
The growth of the algae progressed under sterile conditions. The glass work used was sterilised by heating at 120 degrees for 1 hour; the polythene bottles and the quartz dishes by UV irradiation. The nutrient solution was filtered through a ceramic candle, which was designed as a bacterial filter. All open ends of containers, etc., were either covered or suitably sealed, even when not in use.
7.1.3. Nutrient Solution
The composition given by Kuhl (1962a) was followed with one difference. The magnesium content was reduced to 1/10 of the original stated amount. The missing Mg was replaced by an equimolar solution of Na2SO4. The reason for this reduction was the hypothesis that should a transmutation occur, it would intervene in the first place in the assimilation process, with an increase in the amount of magnesium that is so essential for the production and functioning of chlorophyll. A small increase [supposed Holleman] in the magnesium content could only be determined when the initially given magnesium was as small as possible [see section 10.1.3 for further details].
Otherwise it was demonstrated that the nutrient solution composition made excellent algal growth possible.
The solution was prepared from a few stem solutions (A1-A4, B and C). This was presumably to prevent chemical reactions resulting in the formation of insoluble precipitates. The composition of the stem solutions is given in the table below:
| Kuhl solution [as modified by Holleman] | ||
|---|---|---|
| A1 | KNO3 | 5.5x10-3 |
| KH2PO4 | 4.5x10-3 | |
| NaNO3 | 4.5x10-3 | |
| Na2HPO4.2H2O | 0.5x10-3 | |
| A2 | MgSO4.4H2O | 1.0x10-4 |
| A3 | Na2SO4 | 0.9x10-3 |
| A4 | CaSO4.2H2O | 1.0x10-4 |
| B | FeSO4/EDTA complex | 2.5x10-5 |
| C | H3BO3 | 1.0x10-6 |
| MnSO4.4H2O | 1.0x10-6 | |
| ZnSO4.7H2O | 1.0x10-6 | |
| CuSO4.5H2O | 1.0x10-8 | |
| (NH4)6Mo7O24.4H2O | 1.0x10-8 | |
7.1.4. Algal Cultures
7.1.4.1. Production of Cultures
Cultures of Chlorella vulgaris, originating from pure stock cultures from the Culture Centre of Algae and Protozoa, Cambridge, UK, were kindly made available by the Experimental Physics Laboratory, Section for Spectroscopic Biology, of the Utrecht University.
With the aim of keeping the stock cultures alive for the duration of the whole experiment, they were kept in a dimly lit environment at about 17 degrees. Fresh algal cultures were needed later to prepare each of the six growth periods of the experiment. This was attained, in advance, by means of the gas wash flask, which contained 50ml of nutrient solution inoculated by 2ml of a previously grown inoculation culture, being bubbled through with CO2 rich air and kept at a temperature of approximately 27 degrees. In the inevitable periods of time between the end of growth and its usage for the following cycle, the suspension was stored as stated earlier in this section.
7.1.4.2. Measurement of Cell Number
The number of cells per cm3 in the algal suspension was determined according to the methods of Boney (1974) and Hille (1938). Two millilitres of the algal suspension was centrifuged for 5 minutes at 4000 rpm. The residue was heated with 3-4ml of methanol in a water bath for 5 minutes. After cooling it was centrifuged again and decanted into a 10ml measuring cylinder. The extraction was repeated and decanted after centrifuging into the same measuring cylinder, which was filled at room temperature with methanol to 10ml. The optical density was measured in a spectrophotometer at a wavelength of 660 microns and compared with pure methanol. The reference solution for all following measurements was a chlorophyll extract of the original Chlorella culture obtained following the above prescribed methods. The linear relationship between optical density and concentration of the extract was checked by measuring a number of dilutions of this extract. [This method in fact gives a determination of chlorophyll content rather than relative cell density per se.]
7.1.5. Procedure
7.1.5.1. Growth of Cultures
Fifty millilitres of the sterile nutrient solution was pipetted into each dish, as was 2ml of algal suspension (grown in the same nutrient medium). The suspension was brought over with a 'Stas pipette' or so called 'Swift pipette' to overcome sedimentation during pipetting. The dishes were covered with sterile plastic lids and placed in the shaking bath. To the gas wash flask a culture of the same composition was added. After this was completed, the lids were replaced by the previously described Perspex covers (see 7.1.2.5) which were connected to the gas supply apparatus. The shaking bath was covered and the gas valve opened. The speed of the gas stream (air with 5% CO2) was roughly read from the rate of the bubbles through the gas wash flask.
At the end of a growth period the mineralization of the cultures and control dishes followed.
7.1.5.2. Ashing and the Provision of New Nutrient Solutions
The first step was to adjust the acidity (pH) of the culture solution back to its original value using 10ml of 0.01N nitric acid. This quantity was determined from earlier experiments. The dishes were then placed under an infra red lamp for 5 minutes. During this process the dry remnant was already carbonized and partly ashed. A short heating for 10 minutes in a furnace at 500-520 degrees was necessary for complete ashing. Because of a chemical reaction between the acid phosphates and the nitrates a second quantity (10ml, 0.01N) of nitric acid was added to the dishes, after ashing, at a moderate temperature to neutralise this reaction. Finally the ash was dissolved in twice distilled water to produce 50ml of a new more or less neutral feeding solution. Again, the acidity was checked and determined by means of earlier parallel experiments.
7.1.5.3. Controls
As previously mentioned in 7.1.1, the controls of Holleman's first significant experiment (II) differed from the experimental cultures only in so far as that they were acid treated, evaporated and ashed (7.1.5.2) immediately after the inoculation of the nutrient solution. They were then stored in their original dishes in a desiccating chamber, for the duration of the growth of the experimental cultures, in order to prevent water vapour from affecting the dry ashes. Once the ashing of the experimental cultures was complete, the control dishes were able to be brought out and placed alongside their equivalent experimental dishes for the rest of their parallel treatment and handling.
The treatment of the controls of Holleman's third experiment was a potential improvement on his earlier experiments. The inoculated nutrient solution was treated immediately with the 10ml of 0.01N nitric acid, which for the experimental cultures was added at the end of the growth period prior to evaporation. The idea was that this premature acidification would prevent algal growth. The control dishes were able to remain alongside their experimental counterparts at all times. [Unfortunately the acid proved not to be strong enough to prevent algal growth.]
7.1.5.4. Chemical Analysis
The ashes of one each of the experimental and control dishes were removed in preparation for chemical analysis at the end of each growth cycle. The neutralised ash was dissolved in 5ml 0.01N perchloric acid and briefly (10 minutes) heated over a steam bath. After cooling it was filtered through an ash free filter and/or decanted into a measuring cylinder where it was topped up with twice distilled water to 50ml. The solutions were kept in well closed polythene bottles in the fridge.
Potassium was the first element to be determined. This was done according to a method by Flaschka (1953) by titrimetric measurement of the insoluble potassium salt of tetraphenylborate (Kalignost). The determination was conducted using 5 separate samples taken from each of the 6 experimental and the 6 control ash solutions. Each parallel experimental and control solution was analysed during the same work period. In most cases it was also conducted at the same time as an analysis of the original feeding solution and a calibration solution of an exactly known concentration of 0.01N KCl. (Measured value averaged 1.000x10-2M, n=16, s=0.018x10-2).
Potassium was further determined by flame photometry, as was the other element measured, sodium. The ash solutions were needed to be diluted by 1/100 before analysis.
The fact that sodium was included in the analysis was due to the expectation that it is an unimportant element in the [algal] organic process. It would therefore be a purely neutral part of the feeding solution and should thus strictly follow the conservation law. Assuming this to be true, it was used as an internal standard. Any change in the volume of the culture liquid as a whole, be it through evaporation or pipetting errors, would have no influence on the ratio of the concentration of any element relative to the sodium. Changes in these concentration ratios are therefore a more trustworthy sign of the growth or diminution of an element than would be the concentration itself. [The determinations of sulphate, phosphate, calcium and magnesium proved unsuccessful, the latter pair despite extensive experiments. See section 7.2.3 for details.
7.2. Results
The results for the first significant experiment (II) are given in figure 3, figure 4, figure5 and figure 6.
7.2.1. Analytical Results
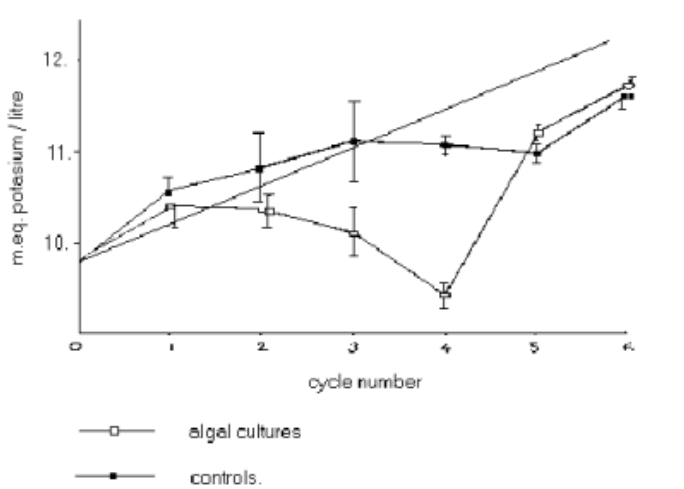
Figure 3: Potassium content of ash solutions by Kalignost method
Figure 3 shows the potassium content of each of the 6 control (solid circles) and 6 experimental (open circles) dishes as measured by the Kalignost method at the end of each of the 6 growth cycles. The error bars associated with each of these values records the range of results obtained from the 5 separate samples taken from the analysed dishes. The dashed line shows the theoretically expected results. The 4% slope of this line is due to the addition, at the beginning of each growth cycle, of an extra 2ml of chemically identical inoculation culture to each 50ml of nutrient solution. The starting point of this line used the experimentally measured potassium content of the pure nutrient solution (average = 0.985x10-2M, n=15, s=0.018x10-2).
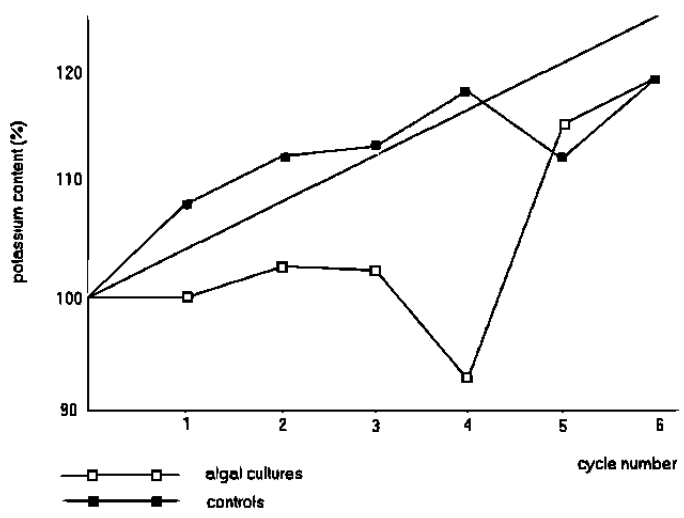
Figure 4: Potassium content of ash solutions by flame photometry
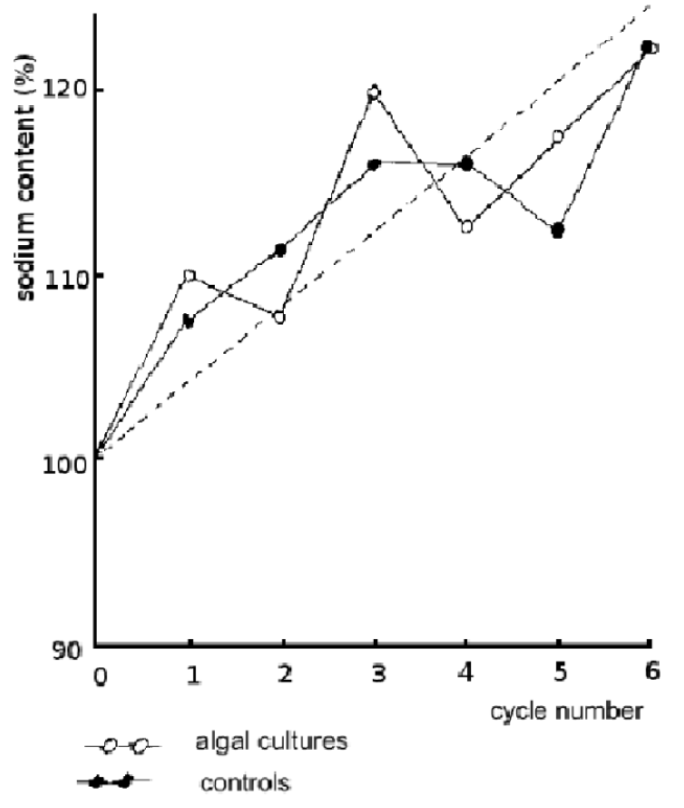
Figure 5: Sodium content of ash solutions by flame photometry
Figure 4 and figure 5 show the results of the potassium and sodium analyses as determined using flame photometry. There are no error bars here as only one sample was taken from each dish for the potassium and sodium analyses.
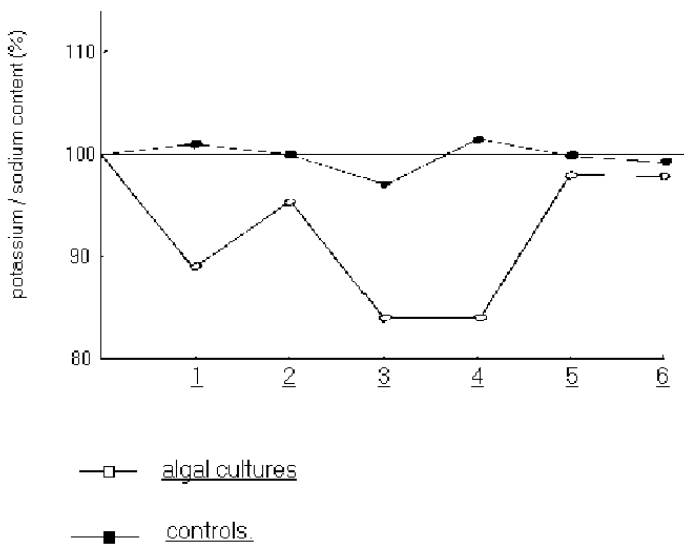
Figure 6: Ratio of potassium to sodium content of ash solutions by flame photometry
Figure 6 shows the ratio of potassium to sodium as measured by flame photometry for each of the control and experimental dishes analysed at the end of each of the 6 growth periods.
The first thing to note is that in comparing figure 3 and figure 4, there is relatively little variation between the two totally independent methods of potassium analysis. Thus any differences observed between controls and experiment must be attributed to factors independent of the final analysis method itself.
A second noteworthy feature is the similarity observed between controls and experiment, as predicted, in the sodium contents, as shown in figure 5.
This contrasts with the relatively large reduction in the potassium content in the experimental cultures, especially for the 4th growth period. This is clearly demonstrated with the potassium/sodium ratios as shown in figure6. The control results have a range of about 4%; the experimental results drop down to a statistically significant 17%.
The return of the potassium content back to expected values for the 5th and 6th growth periods of the experimental dishes remains equally intriguing.
7.2.2. Health of the Cultures
The growth of the algae could be visibly followed in the part experiments by comparing them with the original culture. In the first 3 growth periods it was completely satisfactory, in the last 3 it became less so. Apart from this the appearance of the suspension also changed. Lumps of cells were found and the colour became darker. The same was observed with the inoculation cultures that went with them. The relative quantity [and/or health] of the algae in the inoculation cultures was determined by means of the measurement of their chlorophyll content in comparison with the original culture. It varied between 106-120% in the 6 successive growth periods of 10-14 days.
7.2.3. Sources of Error [Identified by Holleman]
Changes in the volume of the culture solution during the growth phase of the experiment resulting in later analytical errors would include pipetting errors. Of note here is that during the 5th growth phase a reduction in the shaking bath's oscillating frequency resulted in an unexpectedly strong wave in the relevant dishes. Such a loss, Holleman believed, could account, at least in part, for the low analytical values recorded for the part experiments 5 and 6 (figure 3, figure 4 and figure 5). A third source of error was the evaporation which occurred with the ashed solutions being stored awaiting analysis. This was despite their being stored in a fridge in good closing polythene bottles. A small evaporation could not be avoided due to the long storage times determined by circumstance. This could account for the generally slightly high values recorded for the earlier part experiments. [I feel it important to point out that an evaporative loss at any other stage of the experimental procedure other than during ash solution storage would not be detected in the analytical results].
Whilst making the initial nutrient solution a small precipitate was found to occur (despite precautions) which may account for the slightly reduced starting point for the potassium content given in figure 3.
The original plan to determine the mineral content for all cations (see section 6.2) of the nutrient solution could not be realised. The analysis of the next two most important elements, calcium and magnesium failed because of the formation of a precipitate in the ash solutions that did not, or only very slowly, dissolve in mineral acids. It occurred mostly in the experiments in which algae had grown more than three times in one and the same quartz dish. Despite the absence of a definitive identification, on the basis of an article by Hattingberg, et al (1966) it was suspected to be a calcium and/or magnesium polyphosphate salt that accumulated due to the cumulative working method. Possible further references quoted in Holleman's laboratory notebooks include Pascal (1957); Karbe and Jander (1942); Ullman (1921); v. Wazer (1950, '52, '53,'56); Griffith (1956).